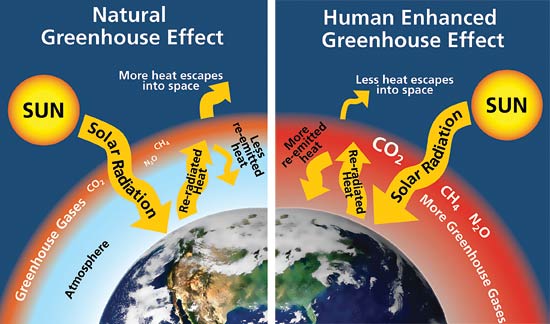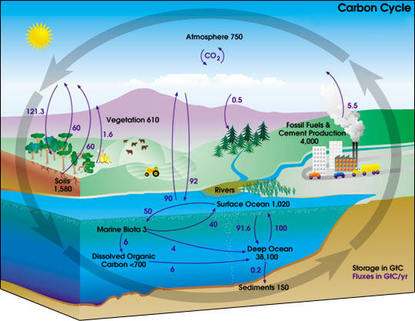
Greenhouse gases are in the ocean in three forms: salute carbon dioxide (CO2), hydrogen carbonate (HCO3), and carbonate (CO3). The most common one is carbon dioxide. When the balance of carbon dioxide in the ocean and air is equal, the exchange process is balanced. Because of the hydrogen carbonate and carbonate being in the ocean, however, it has the capacity to hold more carbon dioxide. The colder the water, the more CO2 is dissolved in it, so in the tropics there is less carbon dioxide in the ocean and more in the air, but in the polar regions there's more CO2 in the water and less in the air.
For photosynthesis to occur, producers need carbon dioxide. Because of the abundance in the ocean, plants like algae can grow faster. This means that algae-eating organisms, such as plankton, can't keep up with the rapid rate of reproduction. The overpopulation of algae uses all the nutrients, and it dies.
The large amounts of carbon dioxide also cause ocean acidification. Nearly a quarter of CO2 released ends up in the ocean, which is equivalent to about 22 million tons a day now. This is the largest observed change in ocean chemistry in the past 50 million years. Scientists haven't been worried about this because they thought buffering would keep it stable. Buffering is when chemicals from rocks flow through rivers into the ocean to stabilize the effect of the acidification. however, recently they have realized that natural buffering can't keep up with the acidification.
Such a rapid change in ocean conditions don't allow for animals to adapt. The shells of some creatures are now dissolving in the more acidic waters. However, scientist have only been studying the biological aspect of this since 2003, when the term "ocean acidification" was first coined. This means that their data is limited to the point that they can't predict the future for the wildlife. Some animals will survive the changes, but others may not. Some may even thrive. But the ones that don't survive will mean a narrowing in biodiversity in the ocean.
This also affects humans, though. The loss in biodiversity means that fisheries and other forms of aquaculture will be affected. This will threaten tourism and other sea-related economies. This means that the people in these industries will be affected, meaning possible food insecurity for millions.
The people rely on the wildlife, and that's where the direct damage from ocean acidification is happening. The small changes in pH, or acidification, can severely affect chemical reactions needed for basic life functions. In marine wildlife these changes impact chemical communication, reproduction, and growth. The skeletons of some marine creatures are sensitive to pH changes. Shell building organisms can't extract the carbonate ion they need in order to grow shells. The acid in the ocean decays coral reef while delaying the growth of new reefs. Plankton are very important in the carbon cycle because they control the greenhouse gas effect between the ocean and the air. Oceans have the largest amount of actively cycled carbon in the world. The oceans are also very important for storing carbon. When shelled plankton (also called phytoplankton) die they bring their calcium carbonate shells to the ocean floor, which then become rock and sediment and can be stored for the future. The plankton reproduce at such a fast rate than other organisms that they can adapt faster and better to the changes in the acidification in the ocean. However, studies show that foraminifera (a type of plankton) do not cope with high acidity levels well because their shells dissolve rapidly. Already, shells of pteropods (another type of plankton) are already dissolving in the Southern Ocean where the more acidic water from the deep sea rose to the surface, hastening the effects of human-derived carbon dioxide acidification. Like coral, the plankton have shells made out of aragonite (a delicate type of calcium carbonate) that is 50% more soluble in water than other types of calcium carbonate. It is unknown how jellyfish will handle acidification.
Plants and algae may thrive in acidic conditions. They make energy through photosynthesis, which uses carbon dioxide, so more in the water is helpful. Seagrasses form shallow water ecosystems along the coasts that are used as nurseries for larger fish that can also be habitats for other organisms. Under acidic conditions, they grow faster and develop deeper roots. However, they are declining for many other reasons, the most prominent being pollution flowing into coastal seawater. This means that the acidification will be unlikely to balance out other factors. Some species of algae grow better in more acidic conditions, but others, such as coralline algae, don't do as well. These species do poorly because they build shells from high-magnesium calcite, which is a form of calcium carbonate that is even more soluble than aragonite or regular calcite forms. A study found that under acidic conditions coralline algae cover 92% less area, making space for other non-calcifying algae, which can damage coral reefs. This is also bad because coral larvae prefer to settle on coralline algae when they are ready to leave the plankton stage.
One major group of plankton, coccolithophores, grow shells. Some studies show that their shells weakened, which makes them more susceptible to damage. A long term study found that a common coccolithophore called Emilania huxley reproduced for 70 generations (it took about 12 months) in the warmer and more acidic conditions. The study showed that the population grew strong shells and adapted to the conditions.
Fish cells will balance with the seawater by absorbing carbonic acid. This changes the pH of the fish's body, which is a condition known as acidosis. Many of the chemical changes that take place in the fish's body may be changed because of this condition. The fish has to put it's body in overdrive to return it to normal conditions. The fish has to use extra energy to excrete the extra acid out of it's blood through its gills, kidneys, and intestines. Burning this extra energy can slow down other things such as growth and digestion. The acid may also affect the fish's minds. For example, clownfish normally hear and avoid noisy predators, but in the more acidic water they do not flee the noise. They also stray farther from their homes and have trouble finding their way back. Scientists hypothesize this because of acidification changing the pH levels in the fish, which affects their brains and bodies. Studies have also found that cobia grow larger otoliths, which are small ear bones that affect hearing and balance. This could affect their ability to navigate and avoid prey. Overall, the ability to adapt to higher acidity will vary between different species of fish.
Scientists have been studying cores, soil, and rock samples that have been taken from the crust dating back to 65 million years ago to see if the carbon dioxide levels and temperature have ever been the same as they are today. They have discovered that the last time Earth has experienced these levels of CO2 was 35 million years ago. This was shown in the chemical composition of fossils in cores from the Earth. 58 million years ago large amounts of carbon dioxide were released into the atmosphere and the temperature rose 9 degrees Fahrenheit (5 degrees Celsius). This is known as the Paleocene-Eocene Thermal Maximum. It's unknown why it happened, but theories include extreme volcanic activity, breakdown of ocean sediments, and widespread fires that burned forests, peat, and coal. The acidification of the water increased rapidly as the carbon dioxide rose. This caused a sudden "dissolution event" where so much shelled sea life disappeared that the sediment went from white calcium carbonate to red-brown mud. 300 million years ago scientists believe that there were changes similar to human-driven acidification, including the near extinction of coral reef. However, no past incident has been the same as what's happening today.
In order to predict what will happen to the natural life, scientists run lab experiments that include placing organisms in tanks of water with raised acid levels and seeing how they react. In these tests, they aren't just looking for shell-building ability, but also for behavior, energy use, immune response, and reproductive success. They also see in what stages of life animals adapt because sometimes the young will adapt and the adults won't, or the adults adapt and the young won't. Studying the effects of stressors such as acidification, higher temperatures, and pollution is important because those are all different ways the humans change the oceans. In the wild, algae, plants, and animals aren't isolated. Instead, they are parts of larger communities of organisms. Some researchers are studying the effects of acidification on the interactions between predators and prey. A snail called the common periwinkle (Lottorina littorea) builds a weaker shell and avoids crabs, but also spends less time looking for food. Boring sponges drill into coral skeletons and scallop shells more quickly. The late stage larvae and black-finned clownfish lose their ability to smell the difference between predators and non-predators. Sometimes they are even attracted to predators.
There are places scattered throughout the ocean where cool carbon dioxide rich water bubbles form volcanic vents. This lowers the pH in the surrounding water, making it more acidic. Scientists can study these unusual communities for clues to what a more acidic ocean would look like. Researchers working off of an Italian coast compare the ability of 79 species of bottom dwelling invertebrates to settle in areas in different distances from CO2 vents. For most species, such as worms, mollusks, and crustaceans, the closer to the vent, and the more acidic the water, the fewer number of animals were able to survive. Animals and algae that need a lot of calcium carbonate were absent or less abundant in acidified water, which were dominated by dense strands of seagrass and brown algae. One species of the polychaete worm (Syllis prolifers) was more abundant in acidic water. The effects of carbon dioxide seeps on the coral reefs and Papua, New Guinea are dramatic. Large boulder corals were replacing complex branching forms, and in some places with sand, gravel, and algae beds. The loss of wildlife means that the oceans' various habitats will no longer provide the diversity we depend on.
One challenge of studying acidification in the lab is that you can only look at a couple species at a time. To study whole ecosystems, and the many other environmental effects, scientists have to do it in the field. The biggest field experiment underway in studying acidification is the Biological Impact of Ocean Acidification (BIOACID). In this experiment, scientists from five European countries built 10 mesocosms, which are really big test tubes 60 feet deep that hold about 15,000 gallons of water, and placed them in the Swedish Gullmar Fjord. After letting plankton and other tiny organisms drift or swim in it, the researchers sealed the tubes, and decreased the pH to 7.8, making it more acidic. This is the expected acidity for 2100. The scientists are waiting to see how they react and whether they can adapt. If this experiment, one of the first of it's kind, is successful, it can be repeated in different areas around the world.
Looking to the future, if the CO2 levels stabilize, the ocean with eventually neutralize itself and the pH will return to normal. This is why past periods with high levels of carbon dioxide show no signs of acidification today. But today, the acidification is rising too quickly and neutralization will take up to a few thousand years, which is too long for the organisms who are being affected now and in the near future.
In 2013 CO2 in the atmosphere passed 400 ppm (parts per million), which is higher than any point in time in the past one million years, possibly even the past 25 million years. The safe level for carbon dioxide is 350 ppm, which we passed in 1988. Without the ocean absorbing it, it would be around 475 ppm. The most logical way to solve this would be reducing carbon emissions by burning fewer fossil fuels and finding more carbon sinks, such as re-growing mangroves, seagrass beds, and marshes, known as "blue carbon". If we did this, then over several years, the CO2 would stabilize again in the atmosphere and ocean. However, even if we stopped emitting carbon right now, the Earth would not immediately neutralize because of the lag of our emissions and when we feel the effects. CO2 typically is in the atmosphere for hundreds of years. In the ocean, the effect is amplified because the acidic ocean water mixes with deep water over a cycle that lasts hundreds of years.
With geoengineering, it is possible that we may be able to create technologies that can help us reduce the atmospheric carbon dioxide or the acidity of the ocean more quickly, without needing to cut carbon emissions very drastically. Geoengineering is anything that would require us to deliberately manipulate planetary systems and the biosphere through the atmosphere, ocean, or other natural systems. The main effect of increasing CO2 that is paramount in people's minds is the warming of the planet. Some geoengineering proposals address this through various methods involving reflecting sunlight with tiny, or large, mirrors in orbit around the Earth. However, this doesn't solve the problem of CO2 in the atmosphere. Another idea is to remove the CO2 from the atmosphere by growing more organisms that use it up- phytoplankton. ADding iron, or other fertilizers, in the ocean would cause man-made blooms in phytoplankton, which in turn would absorb the CO2. When the phytoplankton die, they would take the CO2 with them as they sink down and trap it in the deep sea. However, it is unknown whether or not this would affect the marine food webs that depend on phytoplankton or whether or not this would just cause the deep sea to become more acidic.
Obviously we haven't found a solution to this problem, but there are a few things you can do to help. A major way, of course, is to lower carbon emissions and reducing energy intake in your home by doing things such as recycling, turning off unused lights, biking or walking short distances, or even checking the tire pressure in your car tires can lower gas consumption. And of course, you can educate as many as possible and spread the word about ocean acidification.
For photosynthesis to occur, producers need carbon dioxide. Because of the abundance in the ocean, plants like algae can grow faster. This means that algae-eating organisms, such as plankton, can't keep up with the rapid rate of reproduction. The overpopulation of algae uses all the nutrients, and it dies.
The large amounts of carbon dioxide also cause ocean acidification. Nearly a quarter of CO2 released ends up in the ocean, which is equivalent to about 22 million tons a day now. This is the largest observed change in ocean chemistry in the past 50 million years. Scientists haven't been worried about this because they thought buffering would keep it stable. Buffering is when chemicals from rocks flow through rivers into the ocean to stabilize the effect of the acidification. however, recently they have realized that natural buffering can't keep up with the acidification.
Such a rapid change in ocean conditions don't allow for animals to adapt. The shells of some creatures are now dissolving in the more acidic waters. However, scientist have only been studying the biological aspect of this since 2003, when the term "ocean acidification" was first coined. This means that their data is limited to the point that they can't predict the future for the wildlife. Some animals will survive the changes, but others may not. Some may even thrive. But the ones that don't survive will mean a narrowing in biodiversity in the ocean.
This also affects humans, though. The loss in biodiversity means that fisheries and other forms of aquaculture will be affected. This will threaten tourism and other sea-related economies. This means that the people in these industries will be affected, meaning possible food insecurity for millions.
The people rely on the wildlife, and that's where the direct damage from ocean acidification is happening. The small changes in pH, or acidification, can severely affect chemical reactions needed for basic life functions. In marine wildlife these changes impact chemical communication, reproduction, and growth. The skeletons of some marine creatures are sensitive to pH changes. Shell building organisms can't extract the carbonate ion they need in order to grow shells. The acid in the ocean decays coral reef while delaying the growth of new reefs. Plankton are very important in the carbon cycle because they control the greenhouse gas effect between the ocean and the air. Oceans have the largest amount of actively cycled carbon in the world. The oceans are also very important for storing carbon. When shelled plankton (also called phytoplankton) die they bring their calcium carbonate shells to the ocean floor, which then become rock and sediment and can be stored for the future. The plankton reproduce at such a fast rate than other organisms that they can adapt faster and better to the changes in the acidification in the ocean. However, studies show that foraminifera (a type of plankton) do not cope with high acidity levels well because their shells dissolve rapidly. Already, shells of pteropods (another type of plankton) are already dissolving in the Southern Ocean where the more acidic water from the deep sea rose to the surface, hastening the effects of human-derived carbon dioxide acidification. Like coral, the plankton have shells made out of aragonite (a delicate type of calcium carbonate) that is 50% more soluble in water than other types of calcium carbonate. It is unknown how jellyfish will handle acidification.
Plants and algae may thrive in acidic conditions. They make energy through photosynthesis, which uses carbon dioxide, so more in the water is helpful. Seagrasses form shallow water ecosystems along the coasts that are used as nurseries for larger fish that can also be habitats for other organisms. Under acidic conditions, they grow faster and develop deeper roots. However, they are declining for many other reasons, the most prominent being pollution flowing into coastal seawater. This means that the acidification will be unlikely to balance out other factors. Some species of algae grow better in more acidic conditions, but others, such as coralline algae, don't do as well. These species do poorly because they build shells from high-magnesium calcite, which is a form of calcium carbonate that is even more soluble than aragonite or regular calcite forms. A study found that under acidic conditions coralline algae cover 92% less area, making space for other non-calcifying algae, which can damage coral reefs. This is also bad because coral larvae prefer to settle on coralline algae when they are ready to leave the plankton stage.
One major group of plankton, coccolithophores, grow shells. Some studies show that their shells weakened, which makes them more susceptible to damage. A long term study found that a common coccolithophore called Emilania huxley reproduced for 70 generations (it took about 12 months) in the warmer and more acidic conditions. The study showed that the population grew strong shells and adapted to the conditions.
Fish cells will balance with the seawater by absorbing carbonic acid. This changes the pH of the fish's body, which is a condition known as acidosis. Many of the chemical changes that take place in the fish's body may be changed because of this condition. The fish has to put it's body in overdrive to return it to normal conditions. The fish has to use extra energy to excrete the extra acid out of it's blood through its gills, kidneys, and intestines. Burning this extra energy can slow down other things such as growth and digestion. The acid may also affect the fish's minds. For example, clownfish normally hear and avoid noisy predators, but in the more acidic water they do not flee the noise. They also stray farther from their homes and have trouble finding their way back. Scientists hypothesize this because of acidification changing the pH levels in the fish, which affects their brains and bodies. Studies have also found that cobia grow larger otoliths, which are small ear bones that affect hearing and balance. This could affect their ability to navigate and avoid prey. Overall, the ability to adapt to higher acidity will vary between different species of fish.
Scientists have been studying cores, soil, and rock samples that have been taken from the crust dating back to 65 million years ago to see if the carbon dioxide levels and temperature have ever been the same as they are today. They have discovered that the last time Earth has experienced these levels of CO2 was 35 million years ago. This was shown in the chemical composition of fossils in cores from the Earth. 58 million years ago large amounts of carbon dioxide were released into the atmosphere and the temperature rose 9 degrees Fahrenheit (5 degrees Celsius). This is known as the Paleocene-Eocene Thermal Maximum. It's unknown why it happened, but theories include extreme volcanic activity, breakdown of ocean sediments, and widespread fires that burned forests, peat, and coal. The acidification of the water increased rapidly as the carbon dioxide rose. This caused a sudden "dissolution event" where so much shelled sea life disappeared that the sediment went from white calcium carbonate to red-brown mud. 300 million years ago scientists believe that there were changes similar to human-driven acidification, including the near extinction of coral reef. However, no past incident has been the same as what's happening today.
In order to predict what will happen to the natural life, scientists run lab experiments that include placing organisms in tanks of water with raised acid levels and seeing how they react. In these tests, they aren't just looking for shell-building ability, but also for behavior, energy use, immune response, and reproductive success. They also see in what stages of life animals adapt because sometimes the young will adapt and the adults won't, or the adults adapt and the young won't. Studying the effects of stressors such as acidification, higher temperatures, and pollution is important because those are all different ways the humans change the oceans. In the wild, algae, plants, and animals aren't isolated. Instead, they are parts of larger communities of organisms. Some researchers are studying the effects of acidification on the interactions between predators and prey. A snail called the common periwinkle (Lottorina littorea) builds a weaker shell and avoids crabs, but also spends less time looking for food. Boring sponges drill into coral skeletons and scallop shells more quickly. The late stage larvae and black-finned clownfish lose their ability to smell the difference between predators and non-predators. Sometimes they are even attracted to predators.
There are places scattered throughout the ocean where cool carbon dioxide rich water bubbles form volcanic vents. This lowers the pH in the surrounding water, making it more acidic. Scientists can study these unusual communities for clues to what a more acidic ocean would look like. Researchers working off of an Italian coast compare the ability of 79 species of bottom dwelling invertebrates to settle in areas in different distances from CO2 vents. For most species, such as worms, mollusks, and crustaceans, the closer to the vent, and the more acidic the water, the fewer number of animals were able to survive. Animals and algae that need a lot of calcium carbonate were absent or less abundant in acidified water, which were dominated by dense strands of seagrass and brown algae. One species of the polychaete worm (Syllis prolifers) was more abundant in acidic water. The effects of carbon dioxide seeps on the coral reefs and Papua, New Guinea are dramatic. Large boulder corals were replacing complex branching forms, and in some places with sand, gravel, and algae beds. The loss of wildlife means that the oceans' various habitats will no longer provide the diversity we depend on.
One challenge of studying acidification in the lab is that you can only look at a couple species at a time. To study whole ecosystems, and the many other environmental effects, scientists have to do it in the field. The biggest field experiment underway in studying acidification is the Biological Impact of Ocean Acidification (BIOACID). In this experiment, scientists from five European countries built 10 mesocosms, which are really big test tubes 60 feet deep that hold about 15,000 gallons of water, and placed them in the Swedish Gullmar Fjord. After letting plankton and other tiny organisms drift or swim in it, the researchers sealed the tubes, and decreased the pH to 7.8, making it more acidic. This is the expected acidity for 2100. The scientists are waiting to see how they react and whether they can adapt. If this experiment, one of the first of it's kind, is successful, it can be repeated in different areas around the world.
Looking to the future, if the CO2 levels stabilize, the ocean with eventually neutralize itself and the pH will return to normal. This is why past periods with high levels of carbon dioxide show no signs of acidification today. But today, the acidification is rising too quickly and neutralization will take up to a few thousand years, which is too long for the organisms who are being affected now and in the near future.
In 2013 CO2 in the atmosphere passed 400 ppm (parts per million), which is higher than any point in time in the past one million years, possibly even the past 25 million years. The safe level for carbon dioxide is 350 ppm, which we passed in 1988. Without the ocean absorbing it, it would be around 475 ppm. The most logical way to solve this would be reducing carbon emissions by burning fewer fossil fuels and finding more carbon sinks, such as re-growing mangroves, seagrass beds, and marshes, known as "blue carbon". If we did this, then over several years, the CO2 would stabilize again in the atmosphere and ocean. However, even if we stopped emitting carbon right now, the Earth would not immediately neutralize because of the lag of our emissions and when we feel the effects. CO2 typically is in the atmosphere for hundreds of years. In the ocean, the effect is amplified because the acidic ocean water mixes with deep water over a cycle that lasts hundreds of years.
With geoengineering, it is possible that we may be able to create technologies that can help us reduce the atmospheric carbon dioxide or the acidity of the ocean more quickly, without needing to cut carbon emissions very drastically. Geoengineering is anything that would require us to deliberately manipulate planetary systems and the biosphere through the atmosphere, ocean, or other natural systems. The main effect of increasing CO2 that is paramount in people's minds is the warming of the planet. Some geoengineering proposals address this through various methods involving reflecting sunlight with tiny, or large, mirrors in orbit around the Earth. However, this doesn't solve the problem of CO2 in the atmosphere. Another idea is to remove the CO2 from the atmosphere by growing more organisms that use it up- phytoplankton. ADding iron, or other fertilizers, in the ocean would cause man-made blooms in phytoplankton, which in turn would absorb the CO2. When the phytoplankton die, they would take the CO2 with them as they sink down and trap it in the deep sea. However, it is unknown whether or not this would affect the marine food webs that depend on phytoplankton or whether or not this would just cause the deep sea to become more acidic.
Obviously we haven't found a solution to this problem, but there are a few things you can do to help. A major way, of course, is to lower carbon emissions and reducing energy intake in your home by doing things such as recycling, turning off unused lights, biking or walking short distances, or even checking the tire pressure in your car tires can lower gas consumption. And of course, you can educate as many as possible and spread the word about ocean acidification.